Lp-PLA2 FS test characteristics on BioMajesty® JCA-BM6010/C
- Enzymatic test determining the activity of Lp-PLA2
- Liquid-stable, ready-to-use reagents
- For use in serum, EDTA and heparin plasma
- Wide measuring range up to 2000 U/L
- 2-point calibration with stability of 4 weeks
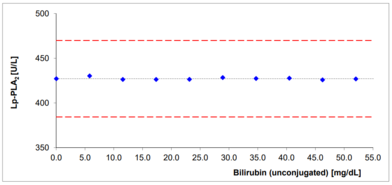
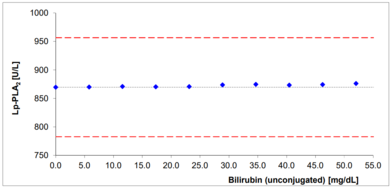
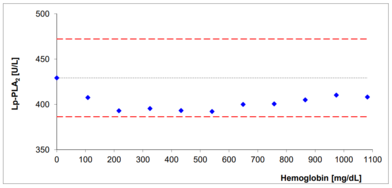
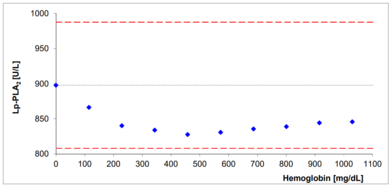
- No significant interferences by ascorbate, lipids, hemoglobin and bilirubin
- Excellent precision over the entire measurement range
- Test is applicable on a wide range of fully automated analyzers
Interferences with bilirubin and hemoglobin
Precision
| Intra-assay n = 20 | Mean [U/L] | SD [U/L] | CV [%] |
|---|---|---|---|
| Sample 1 | 293 | 2.31 | 0.790 |
| Sample 2 | 585 | 3.90 | 0.666 |
| Sample 3 | 848 | 6.56 | 0.774 |
| Total precision CLSI n = 80 | Mean [U/L] | SD [U/L] | CV [%] |
|---|---|---|---|
| Sample 1 | 288 | 7.15 | 2.48 |
| Sample 2 | 572 | 12.3 | 2.15 |
| Sample 3 | 834 | 20.5 | 2.46 |
Method comparison
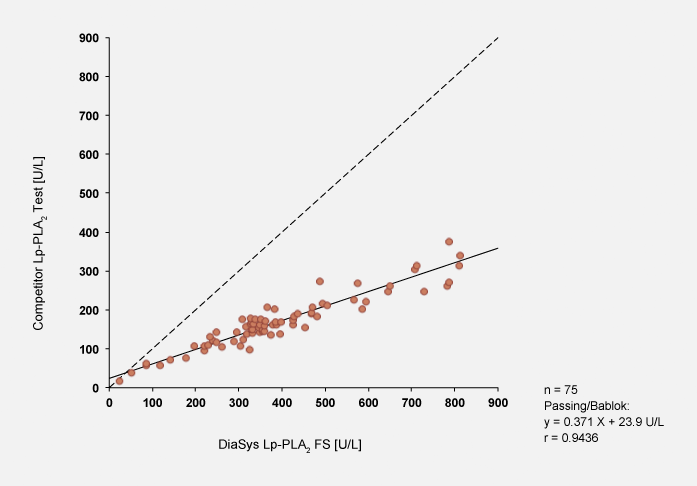
Method comparison was performed against a competitor activity test. Passing/Bablok regression analysis between methods resulted in widely divergent results which may be explained by different standardizations. On average, competitor results are decreased by the factor 2.8. Using the obtained factor, values can be easily converted.